H2S Lesson 1
Hydrogen Sulfide Training
Introduction
Hydrogen Sulfide (H2S) is an invisible, flammable, and toxic gas which is formed from the natural decomposition of decaying plants and animals. Hydrogen Sulfide is very dangerous since it renders people unconscious, paralyzes the respiratory system, and then causes death in just a few minutes. The Bureau of Labor Statistics reported that hydrogen sulfide caused 60 worker deaths between 2001 and 2010.
Hydrogen sulfide is heavier than air so it will collect in low areas. It is water soluble which means that it can be in water and often other liquids such as crude oil. When that liquid is disturbed it can be released into the air.
While Hydrogen Sulfide is known as “rotten egg gas” and it can smell like rotten eggs in low concentrations, it often does not have a smell that can be detected by people.
DO NOT COUNT ON BEING ABLE TO SMELL HYDROGEN SULFIDE!
Hydrogen Sulfide is also corrosive and can cause metal embrittlement. The corrosive effect may appear as sulfide scaling, which can be iron sulfide-a highly flammable substance that can burst into flames when contacting oxygen. When metal is embrittled by Hydrogen Sulfide it can fail without warning.
This class will teach you how to protect yourself and how to react when encountering Hydrogen Sulfide. Despite all the hazards of Hydrogen Sulfide, you can work safely with it being present by;
- Being properly trained

- Planning the job
- Knowing how to react
- Using appropriate PPE
- Using safe work practices
Where could Hydrogen Sulfide Be?
Hydrogen Sulfide is most often produced from the breakdown of vegetable and animal matter, therefore it could be anywhere that plants and animals could be or anywhere their waste could be. Since crude oil is made from plants and animals that have decayed for millions of years, there is often Hydrogen Sulfide in it.
Hydrogen Sulfide can also be produced anytime an acid contacts a sulfur compound. This most commonly happens in a wet cell battery like a truck battery. We need to be especially cautious in confined spaces since Hydrogen Sulfide can become trapped in scales and in the liquids present.
The places we need to be specifically careful are:
- Closed off or confined spaces
- Battery compartments
- Process vessels
- Compressor buildings
- Produce water tanks
- Around oil and chemical tanks
- Wellheads
- Oil spills
- Meter stations
- Pits and trenches
- Dairies
- Breweries
- Fisheries
Since Hydrogen Sulfide dissolves in water workers should expect to encounter it at tank openings, near vents, and around vent lines. When water is heated it can release hydrogen sulfide also.
When scale is removed from tanks using grinding, steaming, or with chemical treatment Hydrogen sulfide can be released into the air. Likewise Hydrogen Sulfide can be in blisters on the sides of tanks.
Characteristics of Hydrogen Sulfide
Some of the characteristics of Hydrogen Sulfide were briefly mentioned in the introduction, however it is important that you gain a better understanding of what it is so we will go into more detail here.
- Hydrogen Sulfide is a colorless and therefore invisible gas. It changes to a liquid at -60C (-76F) so it is unlikely to be encountered in its liquid form.
- Hydrogen Sulfide is heavier than air by about 19%. This means that it will fall into trenches and pits and accumulate there. It is also easily moved by the wind.
- Hydrogen Sulfide is flammable between 4.3% and 46% if an ignition source is available. It will burn with a blue flame and is often burned in flare stacks at refineries and well sites.
- Hydrogen Sulfide is corrosive and when it contacts water hydro-sulfonic acid is created. This can cause metal embrittlement and deteriorate metals with iron content by creating an iron sulfide scale. Additionally hydro-sulfonic acid can irritate the eyes, nose, and throat.
- Hydrogen Sulfide may have a rotten egg smell at low concentrations but since it desensitizes the nerves it will quickly become impossible to smell. It also has no smell at higher concentrations. Never depend on smell for detecting Hydrogen Sulfide.
- Most importantly Hydrogen Sulfide is toxic! It ranks as the second most deadly natural gas after hydrogen cyanide. It kills by paralyzing the involuntary nervous system for the respiratory system. This results in a lack of oxygen to the brain and therefore death.
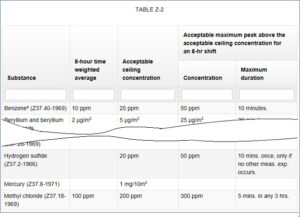
Based off this OSHA and others have published critical limits. The first limit to look for is the Permissible Exposure Limit. Hydrogen Sulfide is often stated as being 10PPM, however this is not correct for General Industry. It is correct for construction and longshoring and marine terminals and it was correct for General Industry on the 1989 rule, but this has changed.
The reason 10PPM is often still used is there is no correct PEL and the rest is a bit complicated. OSHA has decided that 20PPm is OK but only as a ceiling limit unless the person is only exposed for less than 10 minutes in an 8 hour shift then it can be as high as 50PPM (see the excerpt from Table Z-2 of 29 CFR 1910below1910.1000 below).
The National Institute of Occupational Safety and Health (NIOSH) has established a Recommended Exposure Limit (REL) of 10PPM. The American Conference of Governmental Industrial Hygienists (ACGIH®) has set a short term exposure limit (STEL) of 5PPM and a Threshold Limit Value (TLV) of 1PPM. Even with all this confusion, everyone agrees on an Immediate Danger to Life and Health (IDLH) level of 100PPM.